The OPRA™ Implant System is based on 60+ years of science and clinical data. Rooted in the concept of osseointegration, it works by precisely placing a threaded titanium implant into the residual limb where the bone naturally integrates implant to form a strong, lasting anchor to attach a prosthesis.
The prosthesis is connected using the OPRA™ Axor™ II with Guide, creating a secure and quick attachment without the need for a socket. Through personalized rehabilitation, users regain strength and mobility so they can move through life with new energy and confidence.

The OPRA™ Implant System
The OPRA™ Implant System provides direct skeletal anchorage for an external prosthesis. The OPRA™ Implant system is intended for skeletally mature patients.
A natural barrier you can trust with BondedMatrix™
While others have followed, only our design and surgical technique together form the BondedMatrix™ - a unique interface at the skin penetration area that sets the highest standard in integration and stability.
BondedMatrix™ is a biologically integrated barrier that connects the skin to the bone
- stabilizing tissue, minimizing drainage, and promoting a healthy appearance.
Confidence at the point of connection
The Axor™ II with Guide is the only connection device on the market with two planes of release. The Axor™ II releases when subjected to high bending and/or rotational movements to help reduce excessive loads on the OPRA™ Implant System. These are easily resettable by the patient.
The Axor™ II with Guide is used to connect a user’s bone-anchored OPRA™ Implant System with a full-length prosthesis of their choice.
The Axor™ II with Guide allows for donning and doffing to be performed without the need for external tools.
FDA and CE Approved
The OPRA™ Implant System is currently the only FDA-approved bone-anchored prosthetic solution for individuals with transfemoral (above-knee) amputation in the United States.
The OPRA™ Implant System received from FDA Humanitarian Device Exemption (HDE) in 2015 and Premarket Approval (PMA) in 2020 for individuals with above-the-knee (transfemoral) amputation.
The OPRA™ Implant System was CE marked in 1999 and is currently approved under EU Medical Device Regulation (MDR) for transfemoral, transhumeral, and digit amputations. And is also available and approved for use in the UK, Australia, Ukraine and Israel.
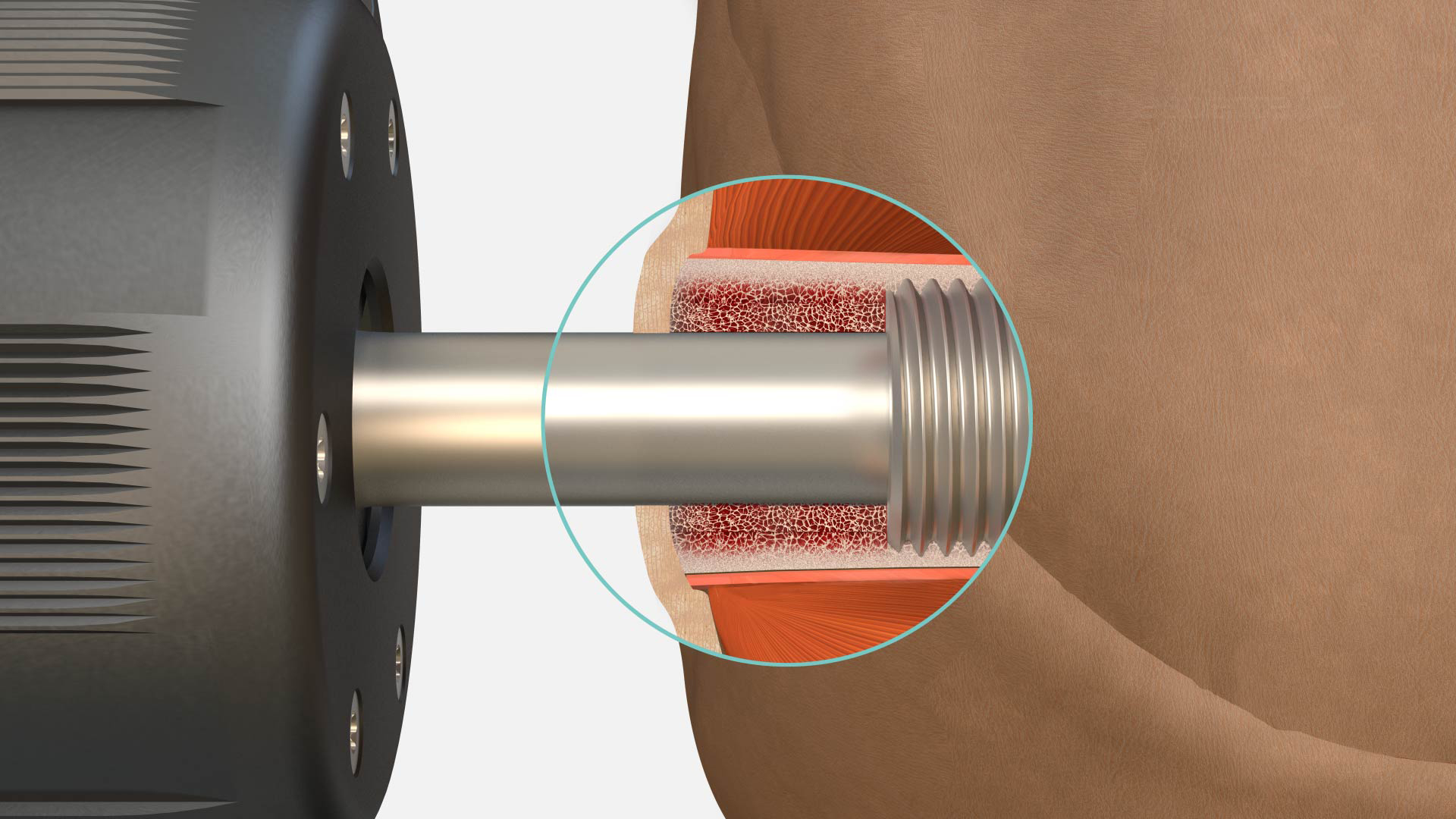
How OPRA™ works
While the product has evolved over the years, and is available in many indications, today’s system consists of three main components: an anchoring element (the Fixture) and a skin penetrating device (the Abutment), secured with a screw (the Abutment Screw). The prosthesis is then directly attached to the external part of the Abutment with an OPRA™ Prosthetic Component.
What is osseointegration?
Osseointegration is a natural phenomenon that has revolutionized the implant industry since the discovery made by Dr. Per-Ingvar Brånemark in the early 1960s.
Dr. Rickard Brånemark, orthopaedic surgeon and Integrum’s founder, expanded upon his father’s pioneering work and applied the principles of osseointegration to rehabilitation for amputees.

Is OPRA™ right for you?
The OPRA™ Implant System is indicated for patients with limb loss due to trauma or cancer who have completed bone growth and currently experience, or are expected to experience, challenges with a socket prosthesis.
Examples of rehabilitation problems:
- Recurrent skin infections and ulcerations in the socket contact area
- Pain
- A short stump preventing the use of socket prosthesis
- Volume fluctuation in the stump
- Soft tissue scarring
- Extensive area of skin grafting
- Socket retention problems due to excessive perspiration
- Restricted mobility
Find a center of excellence
Ready to take the leap? Find a Center of Excellence near you and schedule an initial consultation.